
Abstract : Molybdenum exhibits many advantages in semiconductor industry.Summarized the physicochemical properties, preparation processes,and application directions of three kinds of molybdenum precursors (molybdenum hexafluoride,mo- lybdenum dichloride dioxide,and molybdenum hexacarbonyl).
1 Introduction
Elemental molybdenum possesses numerous advantages, including a high melting point, low thermal expansion coefficient, low bulk resistivity, short mean free path, ease of stress control, high binding energy, and strong adhesion to low-k dielectrics. As a result, it plays an increasingly important role in the semiconductor industry—specifically in the chip manufacturing processes for integrated circuits (including 3D NAND, DRAM, and advanced logic).
Generally, molybdenum metal is used to fabricate various integrated circuits via atomic layer deposition (ALD) or chemical vapor deposition (CVD). However, pure molybdenum, which has a high melting point (2,623°C) and a high boiling point (4,639°C), is difficult to deposit directly; it must first be vaporized as a precursor and then decomposed into elemental molybdenum for deposition. Molybdenum hexafluoride (MoF₆), molybdenum dichloride dioxide (MoO₂Cl₂), and molybdenum hexacarbonyl [Mo(CO)₆] all offer advantages such as low boiling points and controllable decomposition, making them excellent precursors currently used for deposition.
This article will focus on the high-purity synthesis processes for the three precursors mentioned above and their current cutting-edge applications in the semiconductor industry.
2 Preparation and Applications of MoF₆
2.1 Physical and Chemical Properties of MoF6
MoF₆ has a molecular weight of 209.94, a melting point of 17.5°C, and a boiling point of 34°C. At room temperature, it is a volatile liquid (density 2.544 g/mL), and at low temperatures, it forms white crystals (density 2.888 g/cm³). It is highly sensitive to water and readily hydrolyzes to form MoO₃ and HF [1].
2.2 Process for the Preparation of MoF6
O'Donnell et al. [2] reported a synthetic route for MoF₆: in a copper reactor, pure molybdenum was directly fluorinated with fluorine gas (F₂), and the product was collected in a trap cooled to −78°C containing sodium fluoride (NaF), which effectively removed impurities such as HF and SiF₄. The reaction equations are shown in Equations (1) to (3).
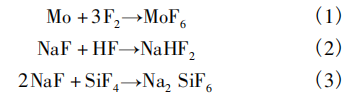
Ghorab [3] reported an optimized process route. The reaction setup consists of: pretreatment trap A containing NaF; a nickel reaction tube containing a nickel boat filled with molybdenum powder; post-treatment trap B containing NaF; and collection trap C containing NaF. The procedure is as follows: First, purge the reaction setup with nitrogen; then NaF is heated and activated under vacuum; nitrogen purging is continued to preheat the reaction tube to 350°C; traps A, B, and C are cooled to −76°C using a dry ice–dichloromethane bath; and a fluorine–nitrogen mixture (F₂:N₂= 1:3) is introduced to initiate the reaction.
Currently, the industrial-scale process for preparing MoF₆ is essentially a scaled-up version of the process described above. The Kanto Denka Kogyo Group in Japan [4] has reported on a MoF₆ production facility characterized by a tiltable fixed bed containing molybdenum powder, which achieves a crude MoF₆ yield of up to 90.3% and a purity of up to 99.9347%.
MoF₆ used in the semiconductor industry requires electronic-grade purity. Purification can be performed by following the method for producing ultra-high-purity tungsten hexafluoride developed by Praxair [5], with the specific process outlined below: 1. Vaporize MoF₆ (at 34°C) to remove heavy metal impurities; 2. Pass the gas through NaF or KF adsorbents to remove HF and SiF₄; 3. Maintain MoF₆ in a liquefied state (slightly above 17.5°C) and perform repeated “helium (He) bubbling–vacuum” cycles under a pressure of 0.38–1.07 bar(g) (Note: 1 bar = 0.1 MPa) to remove light-component impurities such as SF₆, CO₂, CF₄, O₂ + Ar, CO, and N₂.
2.3 Applications of MoF₆
MoF₆ is classified as a corrosive and toxic gas. It must be packaged in DOT-approved cylinders made of carbon steel, stainless steel, or Monel alloy, equipped with a CGA330 cylinder valve [1].
In the 1980s, MoF6 was already being used in the manufacturing process of very large-scale integration (VLSI) chips, where molybdenum was deposited as the gate material for metal-oxide-semiconductor field-effect transistors (MOSFETs) and as the metal interconnect material for back-end-of-line (BEOL) processes. General Electric [6] in the United States and the Hitachi Group [7] in Japan utilized laser-induced chemical vapor deposition of molybdenum to directly write refractory metal lines onto silicon surfaces. The process is as follows: Under conditions of 400°C and a vacuum of 0.5–10 Torr (Note: 1 Torr = 133.32 Pa), an ArF excimer laser (λ = 193 nm) is directed at the silicon wafer surface with an energy of 200 mJ and a repetition rate of 10 Hz, while the feed gases MoF₆ and H₂ (using Ar as the carrier gas) are introduced for deposition. The reaction equation is given by Equation (4).
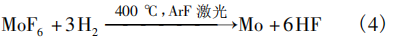
In recent research, the scaling of advanced logic circuit technologies has posed significant challenges for the size and process integration of metal interconnects. Due to the sharp increase in line resistance and via resistance, as well as a decline in stability, the performance of integrated circuits is increasingly limited by metal interconnects. Founta et al. [8] point out that molybdenum’s low bulk resistivity (53.4 nΩ·m) and small mean free path (11 nm) result in a lower effective resistivity, making it a promising candidate for metal interconnect materials that do not require barrier or underlay layers.
3. Preparation and Applications of MoO₂Cl₂
3.1 Physical and Chemical Properties of MoO₂Cl₂
MoO₂Cl₂ has a molecular weight of 198.85, a melting point of 175°C, and sublimes directly at 184°C; at room temperature, it is a yellowish-brown powder (density 3.31 g/cm³). It is slightly soluble in nonpolar solvents and readily soluble in polar solvents. In air, it readily forms a blue (or green) hydrate, MoO₂Cl₂·2H₂O, with water; when dissolved in water, it hydrolyzes to form MoO₃ and hydrochloric acid (HCl aqueous solution).
3.2 Process for the Preparation of MoO₂Cl₂
Colton et al. [9] reported a synthetic route for anhydrous MoO₂Cl₂. The procedure is as follows: 1. Place a porcelain boat containing molybdenum metal into a high-temperature-resistant glass (Pyrex™) reaction tube; 2. Heat to 200°C and purge with N₂ to remove moisture; 3. Continue heating, and at 250–350 °C, introduce a dry oxygen-chlorine mixture (O₂:Cl₂ = 1:1) to initiate the reaction; 4. The volatile products are purified by sublimation and condensation. Additionally, using a similar procedure, MoO₂Cl₂ can be prepared by passing Cl₂ through MoO₃ at 650°C. The reaction equations for the two synthetic routes are shown in Equations (5) and (6).
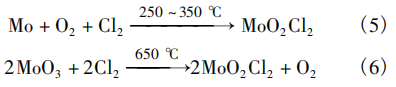
Japan’s JX Metal Group [10] reported a process and apparatus for preparing high-purity MoO₂Cl₂ (5N), with the reaction equation shown in Equation (6). The operating procedure is as follows: 1. Place high-purity MoO₃ powder (4N) in a quartz reactor, introduce N₂ carrier gas, and heat the reactor to 720°C; 2. Maintain the N₂ carrier gas and heating temperature, and introduce dry Cl₂ at a flow rate of 30 mL/min to initiate the reaction; 3. The gaseous reaction products are passed into a collector maintained at 50°C and atmospheric pressure for cooling, where the solid product is sublimated. The yield reached 85.3%, and the purity reached 5N.
Hunan Huajing Powder Materials Co., Ltd. [11] used high-purity molybdenum pentachloride (MoCl₅) as the raw material to prepare high-purity MoO₂Cl₂ (4N) under relatively low-temperature conditions (200–400°C). The process consists of two steps.
1. Preparation of crude MoO₂Cl₂: Place high-purity MoCl₅ (4N) in a tubular reactor, purge with argon (Ar) for protection, and heat the reactor to 200 °C; maintain the heating temperature, shut off the Ar flow, and introduce dry O₂ at a flow rate of 150 mL/min for 4 hours; Shut off the O₂ supply, introduce Ar, and cool the reactor to allow the product to sublimate; collect the resulting solid MoO₂Cl₂ and a small amount of solid MoCl₅. The reaction equation is shown in Equation (7).
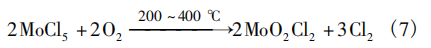
2. Sublimation purification of the crude product: Under an argon atmosphere, the mixture is heated to above 200°C to cause MoO₂Cl₂ to sublimate; the MoO₂Cl₂ vapor is passed through a filter cartridge with 20-nm pores, and the remaining solid impurities are retained in the chamber of the purification apparatus; The MoO₂Cl₂ vapor is directed to a collector where it is cooled and condensed to yield a solid product, with a yield of 95.53% and a purity of 4N.
MoO₂Cl₂ is classified as a corrosive solid. The Japanese company Taiyo Nippon Sanso Corporation [12] has announced a supply technology for high-purity solid-phase precursors of MoO₂Cl₂ used in semiconductors. MoO₂Cl₂ is stored in specially designed containers and used via thermal sublimation.
Since HF etches silicon, MoO₂Cl₂ has garnered significant attention as a fluorine-free precursor. The use of MoO₂Cl₂ in selective molybdenum deposition processes helps reduce the number of steps and costs associated with traditional photolithography, while meeting the demands of semiconductor device miniaturization. Selective deposition in metal-insulator patterns offers significant value in both mid-process (MOL) and back-end-of-line (BEOL) applications.
Lee et al. [13] used MoO₂Cl₂ as a precursor to perform atomic layer deposition (ALD) on the surface of a 12-inch (Note: 1 inch = 25.4 mm) Si(100)/SiO₂(1,000 Å) wafer [i.e., a 100-nm-thick layer of silicon dioxide thermally deposited on the Si(100) crystal plane as a substrate]. The process parameters for depositing the Mo film were as follows: at 600–650 °C under vacuum (11–20 Torr), MoO₂Cl₂ vapor was delivered using Ar as the carrier gas, and H₂ was introduced as the reducing gas for the reaction. The process for depositing the MoN film is similar to the one described above: it requires a lower vacuum (3–5 Torr), a carrier gas flow rate of 100 sccm of Ar, and the introduction of NH3 (10,000 sccm) as the reducing gas for the reaction. The reaction equations are given by Equations (8) and (9), respectively. The MoN film can be used as a diffusion barrier layer for copper interconnects in the back-end-of-line (BEOL) process of integrated circuits.
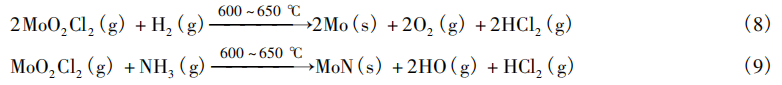
Lam Research Corporation [14] has developed a process for depositing molybdenum at low temperatures using MoO₂Cl₂ on the surface of semiconductor substrates with recessed features. The process is carried out at temperatures ranging from 100 to 500°C, and the procedure is as follows: 1. Expose the semiconductor substrate to silicon-containing reactants, such as silane, ethylsilane, and their various chlorinated derivatives; 2. The semiconductor substrate is exposed to a molybdenum-containing precursor, such as MoO₂Cl₂; 3. The semiconductor substrate is exposed to a reducing agent, such as H₂, NH₃, N₂H₄, B₂H₆, or H₂S; Steps 1–3 are repeated. A key feature of this process is that molybdenum can be deposited non-selectively in trenches, which may consist of a metallic bottom and dielectric sidewalls. This process can be used for the formation of molybdenum silicides in the fabrication of 3D NAND structures, as well as for molybdenum metallization in embedded character line DRAM.
4 Preparation and Applications of Mo(CO)₆
4.1 Physical and Chemical Properties of Mo(CO)₆
Mo(CO)₆ has a molecular weight of 264.01, a melting point of 150°C, and a boiling point of 156°C. At room temperature, it is a white crystalline solid (density 1.96 g/cm³). It is stable in air, insoluble in water, soluble in benzene and paraffin oil, and slightly soluble in organic solvents such as hexane, tetrahydrofuran (THF), and chloroform. Under vacuum, it melts at 142–146°C and decomposes directly at 150°C.
4.2 Synthesis of Mo(CO)₆
Volkov et al. [15] synthesized Mo(CO)₆ by reducing and carbonylating MoCl₅ with zinc (Zn). The reaction was carried out in diethyl ether as the solvent at a CO pressure of 68 bar(g) and room temperature, yielding a Mo(CO)₆ yield of up to 90%. The reaction mechanism is shown in Equation (10).
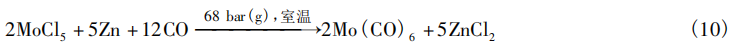
Podall et al. [16–17] reported two methods for synthesizing Mo(CO)₆. The procedure for the first method is as follows: Add benzene and MoCl₅ to a 1 L autoclave; purge and replace the atmosphere twice with CO at 600 psi(g) (Note: 1 psi = 6.89 kPa) [40 bar(g)] to remove air; then introduce CO to a pressure of 1,000 psi(g) [68 bar(g)] and heat to 50°C under stirring; Over the course of 1 h, a solution of triethylaluminum (Et₃Al) is slowly pumped into the reactor under a slight positive pressure of N₂, with the exothermic reaction maintaining the temperature at 65 °C without the need for additional heating; after the addition is complete (Et₃Al:MoCl₅ = 4:1), the mixture is stirred at 65 °C for 2 h. The final Mo(CO)₆ yield was 6%, and the reaction mechanism remains unclear.
The second method uses diethylene glycol dimethyl ether (DG) as the solvent. Under conditions of 900 psi(g) [60 bar(g)] CO pressure and −20°C, MoCl₅ is reduced with sodium (Na) (Na:MoCl₅ = 6:1). The reaction proceeds for 20 h. Subsequently, while maintaining the CO pressure, dilute sulfuric acid is added at temperatures ranging from −10 to 25°C to hydrolyze the intermediate sodium salt Na₂Mo(CO)₅, yielding Mo(CO)₆ with a yield of 65%. The reaction equations are shown in Equations (11) to (13).
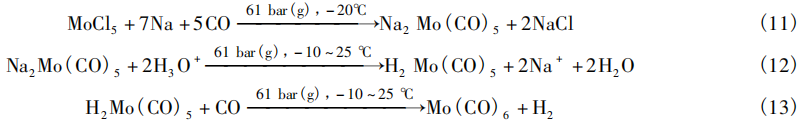
Jilin Jien Nickel Industry Co., Ltd. and the Iron and Steel Research Institute [18] achieved the reduction carbonylation of MoCl₅ at low temperatures using reduced iron powder (Fe) and iron pentacarbonyl [Fe(CO)₅]. The procedure is as follows: Using anhydrous diethyl ether as the solvent, MoCl₅, Fe, and Fe(CO)₅ were added to a reaction vessel in a mass ratio of 1:1:1. Air was first removed by CO displacement three times, followed by the introduction of CO to 120 bar(g). The mixture was then heated to 50°C under stirring; The reaction proceeds for 8 h. After the reaction mixture cools to room temperature, the solvent is removed by distillation, and Mo(CO)₆ is purified by heating to sublimate it, yielding a yield of up to 76%. The reaction equation is shown in Equation (14).
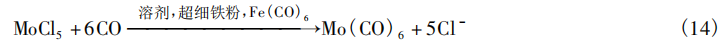
4.3 Applications of Mo(CO)₆
Mo(CO)₆ is classified as a carbonyl metal. At room temperature, Mo(CO)₆ is solid, similar to MoO₂ and Cl₂, and must be stored in specially designed containers; it is used by sublimation through heating. For Mo(CO)₆ packaging, reference can be made to a high-purity W(CO)₆ supply technology provided by Integro Technologies, Inc. [20].
Applied Materials, Inc. [21] has developed a process for the selective deposition of molybdenum using Mo(CO)6 on dielectric and metal surfaces: under vacuum conditions (1–15 Torr) at 150°C, Mo(CO)6 vapor undergoes thermal decomposition directly on the target surface to deposit a molybdenum film. This process is versatile, as it can be performed on both low-k dielectric surfaces (such as SiO₂, SiN, or SiON) and metal surfaces (such as pure metals or their alloys, including Cu, Al, Ti, and W).
Joo et al. [22] employed a plasma-enhanced atomic layer deposition (PEALD) process using Mo(CO)₆ as the precursor to deposit Mo₂N films of controllable thickness on the surface of Si(100)/SiO₂ (1,000 Å) wafers at low deposition temperatures. The process parameters are as follows: at 180–230 °C under vacuum (0.4 Torr), Mo(CO)₆ vapor (generated by bubbling at 40 °C) was delivered using Ar (50 sccm) as the carrier gas and reacted with an electrically excited NH₃ plasma (50 sccm) as the reducing gas. The Mo₂N film deposited at 200 °C is only 0.03 nm thick, making it well-suited for use in scaled-down integrated circuits.
Sebastian et al. [23] used a metal-organic chemical vapor deposition (MOCVD) process with Mo(CO)₆ as the precursor to epitaxially grow a highly crystalline two-dimensional monolayer MoS₂ film on a 2-inch sapphire substrate. The process parameters are as follows: at 1,000 °C under vacuum (50 Torr), Mo(CO)₆ vapor (generated by bubbling at 10 °C under 950 Torr) was delivered using H₂ (0.036 sccm) as the carrier gas, and H₂S (50 sccm) was introduced as the reducing gas for the reaction; After the single-atom-layer growth is complete, the H₂S atmosphere is maintained while cooling to 300°C to prevent decomposition of the MoS₂ film. MoS₂ is a key component of field-effect transistors (FETs), which are currently widely used in various integrated circuits.
5. Conclusion
This paper reviews the research progress on various molybdenum precursors used in the semiconductor industry. As the demand for smaller dimensions in integrated circuits continues to grow, the excellent properties of molybdenum are becoming increasingly apparent, offering considerable application potential. The development of efficient production processes and equipment for electronic-grade molybdenum precursors holds significant strategic importance and practical value.
References: Low Temperature and Specialty Gases ,Vol.44 No.1 Feb.2026;Molybdenum Used in Semiconductor Industry;CHEN Zhiwei,ZHANG Xiaobin,YANG Junhao
Against the backdrop of steady upgrades in the high-end manufacturing sector, a diversified supply of critical materials has become a vital pillar of industry development. Stardust Technology has established a deep presence in the powder materials sector. Building on its core technology of radio-frequency plasma spheronization, the company not only focuses on the R&D and production of rare and refractory metal powders but has also expanded into high-end ceramic spherical powders. By developing comprehensive, multi-scenario powder solutions, Stardust Technology is driving high-quality development across multiple industrial sectors. Leveraging mature core technologies, Stardust Technology’s high-end ceramic spherical powders address practical needs across multiple sectors, including Yb-86 microspheres for targeted cancer therapy, spherical alumina for electronic heat dissipation, and hydroxyapatite for medical aesthetics and orthopedics. Among these, spherical alumina, with its excellent thermal conductivity, efficiently meets the heat dissipation needs of the electronics and information technology sectors. It precisely aligns with the development trends of the 5G and semiconductor industries, providing reliable support for thermal management in products such as high-power chips. The company possesses a professional R&D team and a comprehensive technological framework, with core technologies covering key processes such as powder separation and 3D printing manufacturing. These technologies are highly practical and deliver excellent implementation results. As the lead organization for industry standards, Stardust Technology has spearheaded the formulation of multiple national standards related to additive manufacturing, filling gaps in the domestic industry and laying a solid foundation for its long-term development and the upgrading of industry regulations. Adhering to its positioning of “providing full-industry-chain services supported by core technologies,” Stardust Technology has not only achieved a diversified portfolio of metal and ceramic powders but has also created a three-in-one solution integrating equipment, powders, and services, offering customers one-stop services from R&D and production to validation. Leveraging its solid technical capabilities and proven implementation results, the company continues to provide reliable material support for high-end sectors such as aerospace, biomedicine, and electronics, driving the quality upgrade of “Made in China” through pragmatic innovation. For more product information, please contact our professional sales manager, Cathie Zheng, at +86 13318326187.
