
Abstract: Using MoNbTaW powder prepared by spray granulation as the raw material, spherical MoNbTaW refractory high-entropy alloy powder was produced via radiofrequency plasma. The effects of spheronization power, carrier gas flow rate, and shielding gas composition on the powder spheronization rate were investigated. Scanning electron microscopy (SEM), X-ray diffraction (XRD), laser particle size analysis, Hall flow meters, and nanoindentation systems were used to test and analyze the morphology, phase composition, particle size, flowability, and microhardness of the powder before and after spheronization. The results indicate that the powder did not undergo alloying after ball milling, but after spheronization, it completely transformed into the body-centered cubic (BCC) phase; increasing the plasma power from 32 kW to 40 kW resulted in a higher spheroidization rate, approaching 100%; increasing the carrier gas flow rate from 1 L/min to 4 L/min reduced the number of nanoparticles on the surface of the spheroidized powder, making it smoother, with a spheroidization rate approaching 100%; however, further increasing the flow rate to 7 L/min caused unmelted particles to appear in the powder; adding hydrogen to the shielding gas helped improve the spheroidization rate. After spheroidization treatment, the powder particle size distribution narrowed, the tapped density increased from 2.00 g/cm³ to 8.33 g/cm³, and the bulk density increased from 1.43 g/cm³ to 7.24 g/cm³. the Hall flow time (50 g sample) decreased from 50.8 s to 8.5 s, and the microhardness reached 8.57 GPa.
0 Introduction
In 2010, SENKOV et al. [1] in the United States successfully synthesized two refractory high-entropy alloys with body-centered cubic (BCC) crystal structures—MoNbTaW and MoNbTaWV—for the first time using vacuum arc remelting technology. Due to their excellent high-temperature strength and oxidation resistance, refractory high-entropy alloys hold promise for replacing traditional high-temperature alloys in high-temperature service environments, such as the hot-end components of aircraft engines. Currently, the preparation of refractory high-entropy alloys primarily involves technical approaches such as melting [2], powder metallurgy [3], and additive manufacturing [4]. Among these, the melting method is not only prone to issues such as compositional segregation and coarse grain structure but also typically requires multiple remelting steps, resulting in higher costs. The powder metallurgy method allows for material forming below the melting point, thereby avoiding solidification defects such as segregation. Meanwhile, rapidly developing additive manufacturing technologies, such as powder bed fusion, utilize high-energy beams to rapidly melt and solidify metal powders. The resulting alloys feature fine grains and uniform composition, and since the process does not require molds, it enables near-net-shape manufacturing of complex components. It should be noted that both powder metallurgy and additive manufacturing require high-quality powders as raw materials.
The Radio-frequency Plasma Spheroidization (RFPS) method utilizes the inductive effect of a radio-frequency electromagnetic field to generate plasma, rapidly melting irregularly shaped powder particles and causing them to coalesce into spheres under the action of surface tension, ultimately condensing into spherical powders [5]. Radio-frequency plasma is characterized by high temperature (≥8,000 °C), high enthalpy, controllable atmosphere, and the absence of electrode contamination, making it particularly suitable for the preparation of spherical powders of high-melting-point metals.
Currently, the raw materials used for the preparation of refractory high-entropy alloy powders via radiofrequency plasma spheronization are typically obtained through spray granulation [6] or smelting-crushing [7]. XIA et al. [7] prepared powders with a spheronization rate of 95.3%, a Hall flow rate (50 g) of 15.09 s, an average particle size of 37.5 µm, a bulk density of 7.42 g/cm³, an oxygen content of 0.1677%, and a nanohardness of 7.99 GPa. Although the properties of the prepared powder meet the requirements for 3D printing materials, the preparation process still involves smelting, which is cumbersome and time-consuming, making it unsuitable for large-scale production. However, the fast granulation speed of spray granulation technology can significantly improve production efficiency.
Using spray-granulated MoNbTaW powder as the raw material, the author prepared spherical MoNbTaW refractory high-entropy alloy powder via radiofrequency plasma sputtering. The study investigated the effects of spheroidization treatment on powder properties, as well as the influence of power, carrier gas flow rate, and shielding gas composition on the spheroidization rate of the powder.
1. Experiments
1.1 Experimental Materials
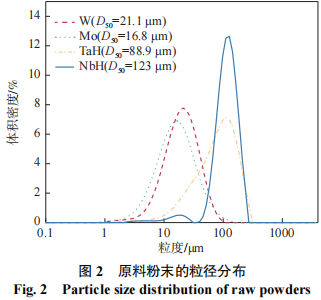
The materials used in the experiments were four types of powders: W, Mo, TaH, and NbH, all with a purity greater than 99.9%. The morphology of the powders is shown in Figure 1. As can be seen in Figure 1, the W and Mo powders are polyhedral in shape, consisting of agglomerated fine particles, while the TaH and NbH powders consist of irregularly broken particles. The particle size distributions of the four powders are shown in Figure 2, with D50 values of 21.1, 16.8, 88.9, and 123 μm, respectively.
1.2 Experimental Procedure
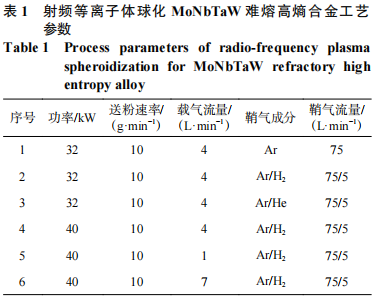
Due to the significant differences in particle size among the four powders, mechanical ball milling was first employed to crush and mix the powders. Using equal molar ratios of W, Mo, TaH, and NbH powders as raw materials, a PTFE ball milling jar and agate grinding balls were utilized. Prior to milling, the jar was evacuated and purged with argon three times to ensure the dry milling process took place in an inert gas atmosphere. An XGB12 planetary ball mill was used. To grind the powders as uniformly and finely as possible within a short timeframe, the ball mill speed was set to the equipment’s maximum of 400 r/min, with a ball-to-powder ratio of 5:1. The dry grinding times were 2, 4, 8, and 12 hours, respectively. After dry grinding, a binder consisting of 1% polyvinyl alcohol (PVA) by mass and deionized water was added for wet grinding, with a powder solid content of 40%. The wet grinding parameters were: speed 300 r/min, ball-to-material ratio 4:1, and grinding time 2.5 h. After mixing, the slurry was pumped via a peristaltic pump into an HF-5L centrifugal spray dryer for spray granulation. The spray granulation parameters were: inlet air temperature 200 °C, outlet air temperature 120 °C, peristaltic pump feed rate 500 mL/h, and atomizer frequency 300 Hz. The granulated powder obtained from spray drying was then fed into the Tekna GN40 radiofrequency plasma spheronization system to prepare spherical WNbMoTa refractory high-entropy alloy powder. The effects of spheronization parameters on powder properties were investigated by varying the plasma power, carrier gas flow rate, and sheath gas composition. Specific process parameters are shown in Table 1.

1.3 Performance Testing
A Zeiss Gemini 300 scanning electron microscope equipped with an Oxford Symmetry S2 EBSD detector was used to observe the surface morphology of the powders and perform electron backscatter diffraction (EBSD) analysis. A Rigaku D/MAX-RC X-ray diffractometer was used to analyze the phases of the powders. The tap density, bulk density, flow properties, and particle size distribution of the powder were determined using a BT-100/300 powder density meter, a BT-200 Hall-effect flowmeter, and a Malvern Mastersizer 3000 laser particle size analyzer, respectively. The microhardness of the powder was measured using an Anton Paar NHT3 nanoindentation system.
2 Results and Discussion
2.1 Preparation of MoNbTaW Refractory High-Entropy Alloy Precursor Powders
Figure 3 and Table 2 show the microstructures of the powders and their D10, D50, and D90 particle size values at different ball-milling times, respectively. After 2 h of ball milling, the powder morphology had undergone significant changes compared to the raw materials. The sharp edges of the relatively brittle TaH and NbH powders had been largely ground away. When mixed with the finer W and Mo powders, the TaH and NbH powders transformed into ellipsoidal particles, and the powder became significantly finer, with a D50 of 5.44 μm. Continuing the ball milling for 4 hours, the powder underwent plastic deformation; the ellipsoidal particles were continuously impacted and compressed by the grinding balls, eventually being crushed into flake-like particles, with the powder D50 decreasing to 4.58 μm. After 8 hours of ball milling, more ellipsoidal particles were crushed into flake-like particles, and the D50 of the powder continued to decrease to 2.75 μm. After 12 hours of ball milling, cold welding occurred in the powder, causing the flake-like particles to fuse together [8] and form lump-like particles; the D50 of the powder increased slightly to 2.88 μm.

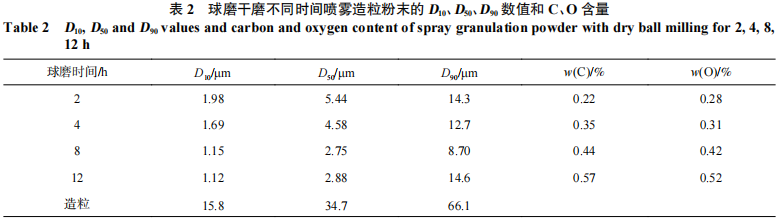
The C and O contents of powders milled for different durations were tested, and the results are shown in Table 2. It can be observed that both the C and O contents of the powders increase with longer milling times. The C primarily originates from the PTFE lining of the ball mill, while the O stems from the fact that as the powder becomes finer during the milling process, its specific surface area increases, causing the surface to adsorb residual O from the wash gas within the ball mill. To ensure uniform elemental distribution in the powder for subsequent granulation and to maintain low C and O content, the powder milled for 8 hours was selected for spray granulation after comprehensive evaluation of powder morphology, particle size distribution, and C and O content.
Figure 4 shows the morphology of the powder obtained from spray granulation. As shown in Figure 4(a), most particles are well-rounded and nearly spherical, but some mushroom-shaped particles and fine powder are also present (as indicated by the arrows). When the slurry is pumped into the granulation equipment, it forms small droplets under the action of the centrifugal atomizer. during which surface water evaporates first, absorbing heat, creating a temperature gradient at the droplet surface. This causes internal PVA particles to migrate toward the surface, and under the influence of gravity, the droplets deform into mushroom-shaped particles. The fragmented powder results from the fact that the rate of PVA diffusion toward the surface is slower than the surface drying rate, leading to an increase in internal water vapor pressure. When the internal pressure exceeds the surface strength, the particles fracture [9–10]. Figure 4(b) clearly shows that the surface of the granulated powder is rough and contains many pores; the powder consists of fine particles and flake-like particles bonded together, which is precisely why the particle size increases and the particle size distribution narrows after granulation. As shown in Table 2, the D50 of the granulated powder is 34.7 μm. EDS surface scanning of the granulated powder revealed that the four elements—W, Mo, Ta, and Nb—are distributed relatively uniformly, with compositions close to the target composition and maintaining nearly equal atomic ratios.
2.2 Effect of Spheronization Parameters on the Spheronized Powder
2.2.1 Effect of Spheronization Power on the Spheronized Powder
Samples from Groups 2 and 4 in Table 1 were selected to study the effect of plasma power on the spheronized powder. During the spheronization process, RF plasma power directly influences the temperature gradient within the plasma torch and the thermal energy of the plasma [11]. At higher power levels, more powder absorbs sufficient heat and is able to melt completely. Figure 5 shows the morphology of MoNbTaW granulated powder at different spheronization powers. It can be clearly observed that the powder spheronization rate increases significantly with increasing plasma power, reaching nearly 100% at 40 kW. In contrast, at lower power (32 kW), the powder contains a significant number of unmelted particles as well as incompletely spheronized particles. It can be inferred that since granulated powders are formed by agglomerating finer particles into near-spherical particles via spray granulation, the high porosity of individual granules results in a large specific surface area; consequently, the 32 kW spheronization power cannot provide the energy required for complete melting of all particles.


2.2.2 Effect of Carrier Gas Flow Rate on Spherical Powder Formation
Figure 6 shows the morphology of MoNbTaW refractory high-entropy alloy powders prepared at different carrier gas flow rates. Under constant spheronization power and shielding gas composition, three different carrier gas flow rates (1, 4, and 7 L/min) were selected to investigate their effects on the spheronization of MoNbTaW granulated powders. At a carrier gas flow rate of 1 L/min, nanoparticles were observed on the surface of the spherical powders. In addition, some irregular powders formed by the adhesion of large and small particles were present, as shown by particles A and B in Figure 6(a). When the carrier gas flow rate was increased to 4 L/min, the number of nanoparticles on the surface of the spherical powder decreased, and the spheronization rate approached 100%; however, distinct “orange peel” patterns appeared on the surface of some particles. When the carrier gas flow rate was increased to 7 L/min, the number of defective powders—such as unmelted particles and satellite particles—increased, and the spheronization effect deteriorated. The experiments indicate that a moderate carrier gas flow rate is crucial for the spheronization of granulated powders. At low carrier gas flow rates, the granulation powder enters the plasma torch at a lower initial velocity, allowing sufficient time to absorb energy and melt completely. At this stage, the probability of small particles colliding and adhering to larger particles increases, making it easier for fine particles to adhere to the surfaces of larger particles. When the carrier gas flow rate is increased to an appropriate value, the optimal initial velocity ensures excellent dispersion of the powder within the plasma torch, resulting in powder with high sphericity and a smooth surface. If the carrier gas flow rate continues to increase, the initial velocity of the powder rises, and the residence time in the plasma torch decreases. Furthermore, a non-conductive region exists between the discharge zone of the plasma torch and the ceramic tube [12], resulting in unmelted particles that leave the high-temperature zone of the plasma torch without passing through it or absorbing sufficient heat. Additionally, higher initial velocities increase collisions between powder particles, leading to the formation of defective powders such as satellite particles.

2.2.3 Effect of Shield Gas Composition on Powder Spheroidization
In the experiments, pure argon (Ar), an argon/helium (Ar/He) mixture, and an argon/hydrogen (Ar/H₂) mixture were selected as shield gases; the spheroidization results are shown in Figure 7. As shown in Figure 7, when pure argon was used as the shielding gas, a large number of unmelted particles and some incompletely melted particles were present, resulting in a low spheroidization rate. Adding helium to the shielding gas effectively improved the powder spheroidization rate, but a small number of unmelted particles still remained. In contrast, when an argon/hydrogen mixture was used as the shielding gas, the spheroidization rate was nearly 100%.

Due to the differences in the thermal properties of argon (Ar), hydrogen (H₂), and helium (He), the amount of energy transferred from the plasma to the powder when these gases are used as sheath gases also varies. The thermal properties of a gas can be quantified using the thermal conductance, which is given by Equation (1) [12].
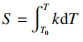 (1)
(1)
In the equation, k represents the thermal conductivity (W/m·K); T₀ is the room temperature, 300 K; and T is the temperature transferred between the powder particles and the plasma, in K.
The energy transferred from the plasma to the powder can be expressed as:
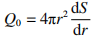 (2)
(2)
In the equation, S represents the thermal conductivity; r represents the average particle size of the powder, in micrometers.
The thermal conductivity of hydrogen is higher than that of helium, and both are significantly higher than that of argon [13]. As shown in Equations (1) and (2), adding hydrogen and helium—which have higher thermal conductivities—to argon increases the amount of energy transferred to the powder per unit time. Consequently, the same mass of powder receives a greater amount of energy within the same time frame, enabling complete spheroidization and resulting in an improved spheroidization rate.
2.3 Analysis of Spheroidized Powder Properties
2.3.1 Analysis of the Physical and Chemical Properties of Spheronized Powders
By observing the spheronization results of the powders in each experimental group, it was found that the best spheronization results were achieved when the plasma power was 40 kW, the powder feed rate was 10 g/min, the carrier gas flow rate was 4 L/min, and the shielding gas atmosphere was Ar/H₂ (flow rate 75/5 L/min). The characteristics of the spheronized powders in this group were analyzed.
First, the particle size distribution of the spherical powder prepared using this spheronization process was analyzed. The spheroidized powder had a D10 of 10.4 μm, a D50 of 21.8 μm, and a D90 of 51.3 μm. Compared to the granulated powder (D10 = 15.8 μm, D50 = 34.7 μm, D90 = 66.1 μm), the D10, D50, and D90 of the spheronized powder were significantly reduced, and the particle size distribution became narrower. This is because during the spheronization process, the binder volatilizes at high temperatures, creating voids within the powder. Simultaneously, the powder absorbs energy and melts, and as it falls, surface tension causes it to condense into spherical particles.
Figure 8 shows the XRD patterns of the powders before and after radiofrequency plasma spheroidization. As shown in Figure 8, the raw powders did not undergo alloying after ball milling. Like the ball-milled powders, the granulated powders exhibit only diffraction peaks corresponding to the individual elemental powders, demonstrating that the granulated powders were formed solely by the agglomeration of the four raw powders under the action of a binder, without any phase transformation leading to the formation of new phases. After spheroidization, the granulated powder completely transformed into the body-centered cubic (BCC) phase. Furthermore, a shift of the diffraction peaks to the left was observed following spheroidization. This is attributed to the solid solution of Mo, Nb, and Ta into the W lattice; since the atomic radius of W is smaller than that of Mo, Nb, and Ta, lattice distortion occurred upon solid solution, resulting in an increase in the lattice constant of W. Based on XRD diffraction data, the lattice constant was calculated to be 0.3211 nm, which is greater than the lattice constant of W (0.3165 nm). The calculated grain size of the BCC solid solution after spheroidization was 18.2 nm.
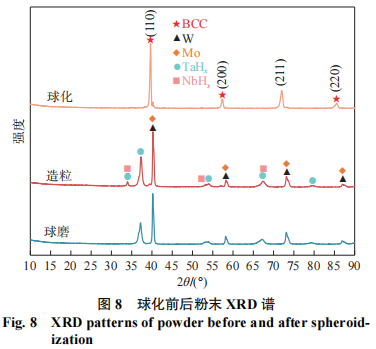
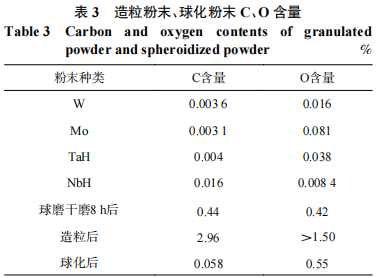
Table 3 shows the C and O contents of the four raw material powders, the powder after 8 hours of dry ball milling, the granulated powder, and the spheroidized powder. The increase in C content in the powder after 8 hours of ball milling is attributed to the PTFE lining of the milling vessel, while the increase in O content is due to residual oxygen remaining after purging the vessel. The C and O contents increased sharply after granulation, primarily due to the addition of the binder PVA. The C and O contents decreased significantly after spheronization; however, the O content after spheronization was 0.1 percentage points higher than that of the powder after 8 h of dry ball milling. This indicates that the PVA did not decompose completely after spheronization and that residues remained. The C content after spheronization was lower than that of the powder after 8 h of dry ball milling because the PTFE introduced during the ball milling process decomposed and volatilized under the high temperatures of the spheronization process.
Test results indicate that the flowability of the powder significantly improved after radio-frequency plasma spheronization, with the Hall flow time (using a 50g timer) decreasing from 50.8 seconds to 8.5 seconds. Although the granulated powder consists of nearly spherical particles, their surfaces are rough and uneven, causing the particles to interlock easily and form bridging, which results in poor powder flowability. The spheronized powder has a smooth surface, eliminating bridging and improving flowability. Additionally, both bulk density and tapped density have increased, bulk density increased from 1.43 g/cm³ to 7.24 g/cm³, and tap density increased from 2.00 g/cm³ to 8.33 g/cm³. This is because the spheronized powder exhibits higher sphericity, reducing the contact area between particles and minimizing the voids in the powder pile, thereby improving both bulk and tap densities.
2.3.2 Microstructure of Spheroidized Powders
Figure 9 shows the cross-sectional SEM morphologies and electron backscatter diffraction (EBSD) patterns of two spheroidized powders of different sizes. One particle has a diameter of approximately 80 μm, and it can be observed that this particle primarily forms dendritic crystals. The other particle has a diameter of approximately 130 μm, and its microstructure consists of equiaxed crystals. The mechanisms underlying the microstructural formation differ between the two particle sizes. After the smaller particle leaves the high-temperature zone of the molten pool, its surface temperature drops rapidly while the interior remains at a higher temperature, creating a significant temperature gradient. Due to its smaller diameter, the central liquid phase does not have sufficient time to undergo supercooling; consequently, nucleation and growth begin at the surface and rapidly extend inward, forming dendritic crystals. For larger particles, after melting is complete, the liquid phase on the surface first exchanges heat with the surrounding environment, resulting in a higher degree of supercooling. This leads to the formation of fine equiaxed grains near the surface. Because the particle size is larger, the liquid phase at the center of the powder has sufficient time to reach a state of supercooling and dissipate heat outward. Crystal nuclei within the interior grow freely in all directions, and due to the limited number of nuclei, coarser equiaxed grains are formed.
During the spheroidization process, some particles remained in the molten pool for a short time and solidified rapidly after leaving it, failing to achieve complete alloying. As shown in Figure 10, unalloyed particles are present on the powder surface. EDS analysis was performed on locations 1, 2, and 3 of the powder; location 1 had a W content of 100%; at location 2, the W content was 87.65%, Nb content was 2.25%, and Ta content was 10.10%; at location 3, the Mo content was the highest at 32.84%, followed by Nb and Ta, while the W content was the lowest at 9.64%. This indicates that during the alloying process, Mo, Nb, and Ta diffuse into W and form a solid solution, which is consistent with the XRD results. The same pattern is observed during mechanical alloying: the lower the melting point of an element, the weaker the interatomic bonding strength, the higher the self-diffusion coefficient, and the easier it is for the element to diffuse into and form a solid solution with elements having higher melting points [14].


The process of alloying granulated powder via radiofrequency plasma spheroidization is shown in Figure 11. The granulated powder consists of nearly spherical particles formed by the agglomeration of four types of irregular powders—W, Mo, NbH, and TaH—under the action of a binder. These particles pass through the high-temperature zone of the radiofrequency plasma torch, where the four elements melt, and then rapidly solidify upon exiting the zone. At the onset of solidification, the lower-melting-point elements Mo, Nb, and Ta begin to diffuse. during this stage, these three elements do not diffuse into the W matrix; however, after a brief period, Mo, Nb, and Ta diffuse and solid-solve into the W matrix. Ultimately, all three elements—Mo, Nb, and Ta—diffuse completely into the W matrix, forming a body-centered cubic (BCC) solid solution, achieving full alloying, and resulting in a refractory, high-entropy MoNb-TaW alloy powder.
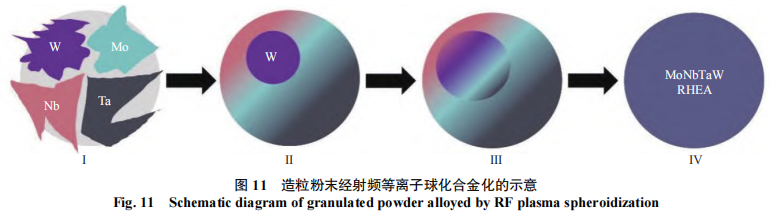
Figure 12(a) shows the cross-sectional SEM morphologies of the granulated powder and the fully alloyed spheroidized powder. It is clearly observed that the granulated powder has a sparse, porous structure, whereas the fully alloyed spheroidized powder is dense and free of voids, as shown in Figure 12(b). This confirms that the narrowing of the particle size distribution after spheroidization is due to the granulated powder shrinking and condensing into spherical particles. An EDS elemental mapping scan of this powder cross-section reveals that the elements are distributed relatively uniformly across individual particles, with no significant compositional segregation. The EDS analysis results indicate that the atomic ratios of the elements are largely maintained, and the elemental content of the spheroidized powder remains essentially consistent with the target composition.
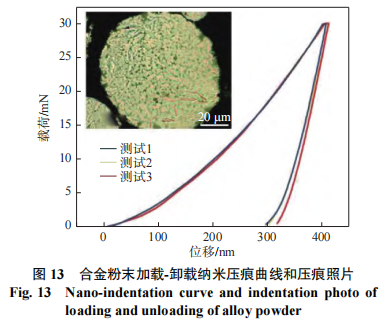
Figure 13 shows the load-unload curves from three nanoindentation tests performed on a single spheroidized powder particle to characterize its mechanical properties. The figure includes a photograph of the indentation after unloading in Test 1, showing a very small triangular indentation at the center of the powder. The microhardness was calculated using the Oliver-Pharr method [15]. The average of the three test results yielded a microhardness value of 8570 MPa ± 119 MPa. Microhardness values obtained by nanoindentation are typically 10% to 30% higher than those obtained by the Vickers method [16]. Taking the upper limit of 30%, the microhardness of the powder is 6 GPa, a value far exceeding that of the fused MoNbTaW refractory high-entropy alloy [2] (4,455 MPa ± 185 MPa). According to the Hall-Pech formula, this is attributed to the finer grain size of the spheroidized powder, which is 18.2 nm (compared to 200 μm for the cast alloy), i.e., grain refinement strengthening.

3. Conclusions
1) Using ball milling parameters of 400 rpm, a ball-to-material ratio of 5:1, and a milling time of 8 hours, and preparing a slurry with 40% solid content and 1% polyvinyl alcohol, it is possible to successfully produce near-spherical granules as precursor powders for radiofrequency plasma spheroidization.
2) By controlling a single variable—either the shielding gas composition, sputtering power, or carrier gas flow rate—the effects of these three parameters on the sputtering outcome were investigated. The final results indicate that the optimal sputtering conditions are achieved at a sputtering power of 40 kW, a carrier gas flow rate of 4 L/min, and a shielding gas composition of Ar/H₂ (flow rates of 75/5 L/min).
3) A comparative analysis was conducted on the properties of powders prepared under optimal spheroidization parameters and granulated powders. Neither the ball-milled nor the granulated powders exhibited alloying; however, after radio-frequency plasma spheroidization, the powders formed a refractory high-entropy alloy with a body-centered cubic (BCC) solid solution structure. After spheroidization, the particle size distribution narrowed, the tapped density increased from 2.00 g/cm³ to 8.33 g/cm³, the bulk density increased from 1.43 g/cm³ to 7.24 g/cm³, and the Hall flow time (50 g sample) decreased from 50.8 s to 8.5 s. After spheronization, powders of different sizes form distinct microstructural patterns. Fully alloyed powders exhibit a dense, pore-free interior with uniform element distribution and no segregation. The spheronized powders possess high microhardness, reaching 8570 MPa ± 119 MPa.
4) The next phase of work will involve using the spheroidized powder for selective laser melting (SLM) to verify the powder’s performance. The focus will be on investigating the effects of laser power and scanning speed on material forming quality, identifying optimal printing parameters, and providing valuable references for the additive manufacturing and application promotion of refractory high-entropy alloy materials. Furthermore, research and development efforts should be intensified to optimize the preparation process parameters for refractory high-entropy alloy powders, ensuring consistent quality and quantity in production. A rigorous quality control system should be established to guarantee the consistency and reliability of the powders, thereby meeting the stringent requirements of high-end application fields.
References: Steel, Vanadium, and Titanium, Vol. 46, No. 2, April 2025; Study on the Preparation of Spherical MoNbTaW Refractory High-Entropy Alloy Powders via Radio Frequency Plasma; Zhao Yumin, Shi Qi, Liu Binbin, Tan Chong, Liu Xin, Zhou Ge, Ding Zhongyao, Qin Feng
Stardust Technology’s core mission is to “become a world-leading supplier of high-end powders, with radio-frequency plasma spheronization technology at its core.” Rather than focusing solely on product sales, the company places greater emphasis on providing end-to-end services, creating integrated solutions that span from powder production equipment and R&D to printing validation. Its ultimate goal is to advance the industrial application of rare and refractory metal materials, providing critical material support for high-end manufacturing sectors such as aerospace and biomedicine. RF plasma spheronization technology represents Stardust Technology’s core competitive advantage. Its principle is simple yet practical: it utilizes ultra-high-temperature plasma to melt irregular raw powder materials, which are then cooled to achieve spheronization, densification, and alloying. This technology effectively addresses industry pain points in traditional powder production—such as low sphericity, insufficient purity, and uncontrollable particle size—and offers significant advantages. Leveraging this core technology, the powders produced by Stardust Technology not only feature high sphericity, minimal internal defects, and flexible particle size control, but are also compatible with a wide range of raw materials, offering outstanding value for money. The company can stably produce spherical powders of various high-end rare refractory metals, including tungsten, molybdenum, tantalum, and niobium, precisely meeting the demands of high-end applications across multiple sectors. With core technology as its foundation and professional service capabilities, the company has established a unique competitive advantage. This ensures the consistent quality of its powder products while providing customers with customized solutions tailored to their specific needs. Stardust Technology remains committed to a pragmatic philosophy, empowering the industry through technology and earning recognition through service, continuously driving high-quality development in the high-end manufacturing sector. For more product information, please contact our professional sales manager, Mr. Duan, at 13378621675.
